Carbohydrates
Episode #1 of the course The molecular building blocks of life by Dr. Bill Thomas
Greetings! My name is Dr. William Thomas, and in this course, I’ll be taking you on a guided tour of the world of biological molecules. As a molecular biologist, I know that many terms scientists take for granted are often poorly understood by the general public, even though they’re used quite frequently. The goal of this course is to de-mystify some of the most important molecular building blocks of life, many of which are part of our daily vocabulary.
Today, we’ll begin with the common carbohydrate. Carbohydrates, often referred to simply as sugars, are usually thought of as food; however, they are also one of the most fundamental building blocks of life. Not only can carbohydrates be broken down through the process of metabolism to provide organisms with the energy they need, but they can be synthesized into more complex molecules for energy storage or for use as structural components. Sugar molecules are even incorporated into DNA!
What Is a Carbohydrate?
While there are many technical definitions for carbohydrates, we can generally think of them as any molecule consisting only of atoms of carbon, oxygen, and hydrogen. The carbohydrates that we typically think of as sugars contain three to six carbon atoms; typically, a sugar molecule will have an equal number of carbon and oxygen atoms and as many hydrogen atoms as oxygen and carbon combined. Now that we have the components of a sugar molecule, how do we put them together?
Carbohydrate Form and Function
A single sugar molecule may be in the form of an open chain or a ring of carbon atoms; in either case, the oxygen and hydrogen atoms can be arranged in multiple ways, making it possible to form different sugars from molecules with the same number of carbon, oxygen, and hydrogen atoms. For instance, glucose, fructose, and galactose are all six-carbon sugars with the chemical formula C6H12O6, yet they are distinctly different. These three sugars are examples of monosaccharides—that is, sugars that consist of one carbohydrate ring.

Glucose, a monosaccharide
Individual monosaccharides can be joined together to form disaccharides, sugars that consist of two rings. Disaccharides with different properties can be obtained from combinations of different monosaccharides. For example, lactose, the sugar associated with milk, is a disaccharide of galactose and glucose, while sucrose, or common table sugar, is a disaccharide consisting of fructose and glucose.
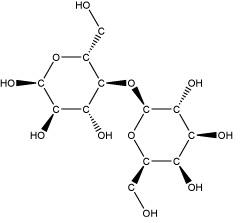
Lactose, a disaccharide produced by linking glucose and galactose
The Growing Chain
Many monosaccharide molecules can be joined together to form a polysaccharide; practically speaking, there is no upper limit to the number of monosaccharide rings that can be contained in a polysaccharide. Just as the oxygen and hydrogen atoms can be arranged differently on a monosaccharide to form carbohydrates with different properties, monosaccharides themselves can be joined together in different arrangements to obtain polysaccharides with different properties. In this way, glucose can be joined together in long chains to form starch, a sugar used by plants to store energy, or in long branching chains to form glycogen, which animals use to store energy.
Joined in yet another way, glucose molecules form the long-chain molecule, cellulose, which is the structural component of plant cell walls. Interestingly, cellulose and other plant-based carbohydrates are formed through the process of photosynthesis, which allows plants to harness the sun’s energy to make glucose out of carbon dioxide and water. This means that the most abundant carbohydrates on earth are made from a recipe for which the only ingredients are sunlight, water, and air!
In the next lesson, we will see how carbohydrates are incorporated into nucleotides, the building blocks of DNA and RNA and the alphabet of the genetic code.
Recommended book
Life’s Ratchet: How Molecular Machines Extract Order from Chaos by Peter M. Hoffmann
Share with friends